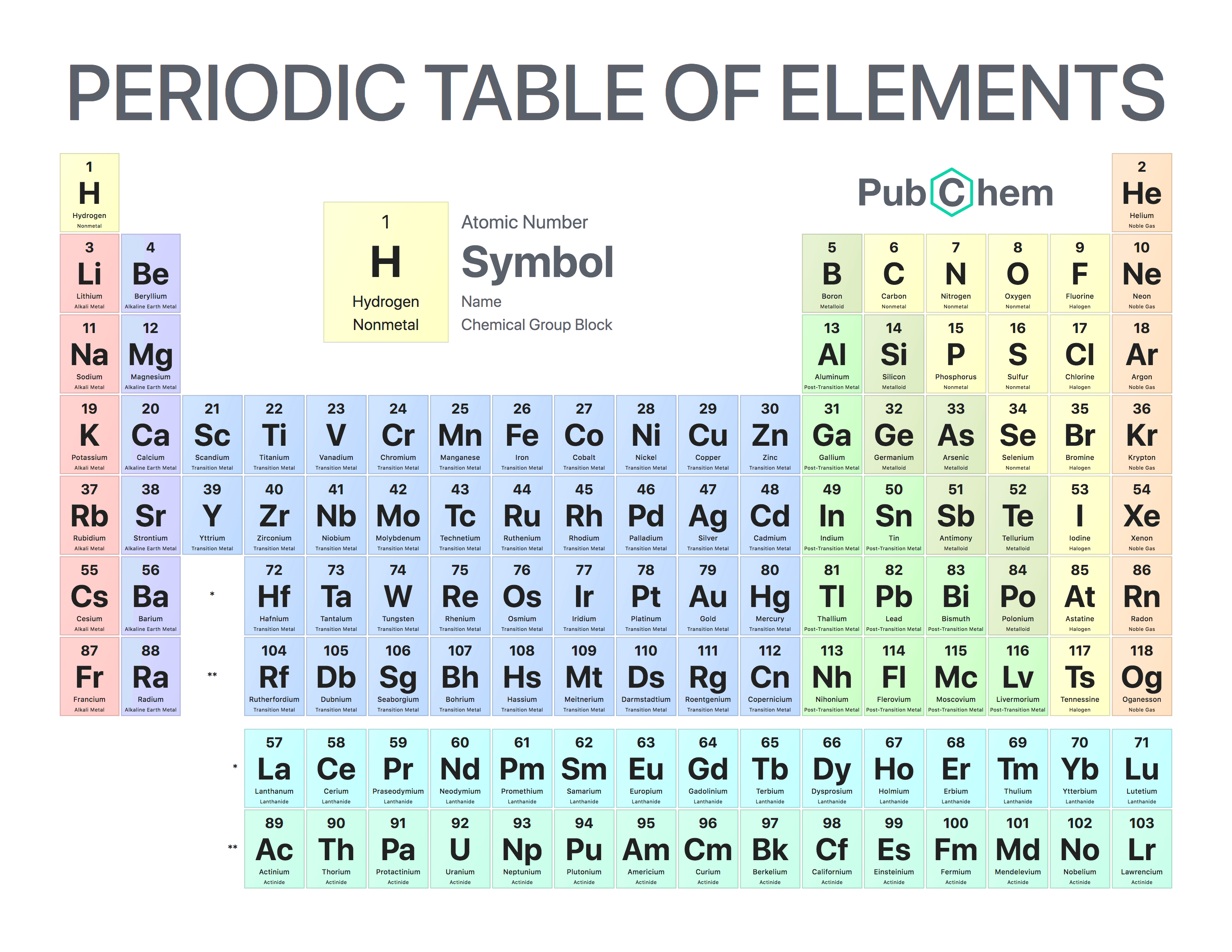
So what is a period on the periodic table? Periods are the horizontal rows of the periodic table. There are 18 groups, and there are 7 periods plus the lanthanides and actinides. Groups are the columns of the periodic table, and periods are the rows. For a more in-depth explanation of periodic trends, click here. These trends tell you where the highest and lowest types of properties are concentrated on the periodic table. A quick way to understand an element’s chemical and physical properties is to know the periodic trends. It is important to note how the location of elements on this table tells us about their properties. From this deduction, he formed the periodic table. In 1869 Russian chemist Dmitri Mendeleev noticed there existed an innate pattern of organization for the chemical elements. Meanwhile, elements in the same period have the same number of occupied electron shells. Elements in the same group have the same number of valence electrons. The periodic table is organized into groups (vertical columns), periods (horizontal rows), and families (groups of elements that are similar). 
You can view all sorts of trends, properties, magnetism, electrons, and even articles on all the elements! The Periodic Table and the Periodic Trends We think our periodic table is one of the best in the world! Visit our new interactive periodic table. View trends, families & groups interactively! Families: Elements that have the same number of valence electrons and therefore similar properties.Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element. 
Groups: The vertical column of the periodic table that signifies the number of valence electrons in an element.Elements: A pure substance composed of a single atom with a unique atomic number.How to Write Electron Shell Configurations.If you enjoy this article, be sure to check out our others! Related Articles: In addition, you will learn about the different properties of the periodic table groups, periods, and families. We will take a close look at the groups of the periodic table. As expected, semimetals exhibit properties intermediate between metals and nonmetals.In this tutorial, you will learn how to read the periodic table. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
